|
Ensuring Validity of Results: This section emphasizes using valid methods, proper sample handling, and traceability to national and international standards for accurate and reliable test results.Process Approach: ISO 17025 employs a process-oriented approach, encouraging laboratories to identify, define, and monitor key testing and calibration processes.Resource Management: It highlights the significance of suitable facilities, equipment, calibration standards, and a conducive working environment.Structural Requirements: ISO 17025 emphasizes the importance of a robust organizational structure, ensuring efficient resource allocation and competent personnel for specific tasks.General Requirements: This section addresses the management and organization of the laboratory, including the establishment of a quality management system, defining roles, responsibilities, and implementing procedures.By adhering to ISO 17025, laboratories enhance credibility, foster customer trust, and ensure compliance with global quality assurance practices. 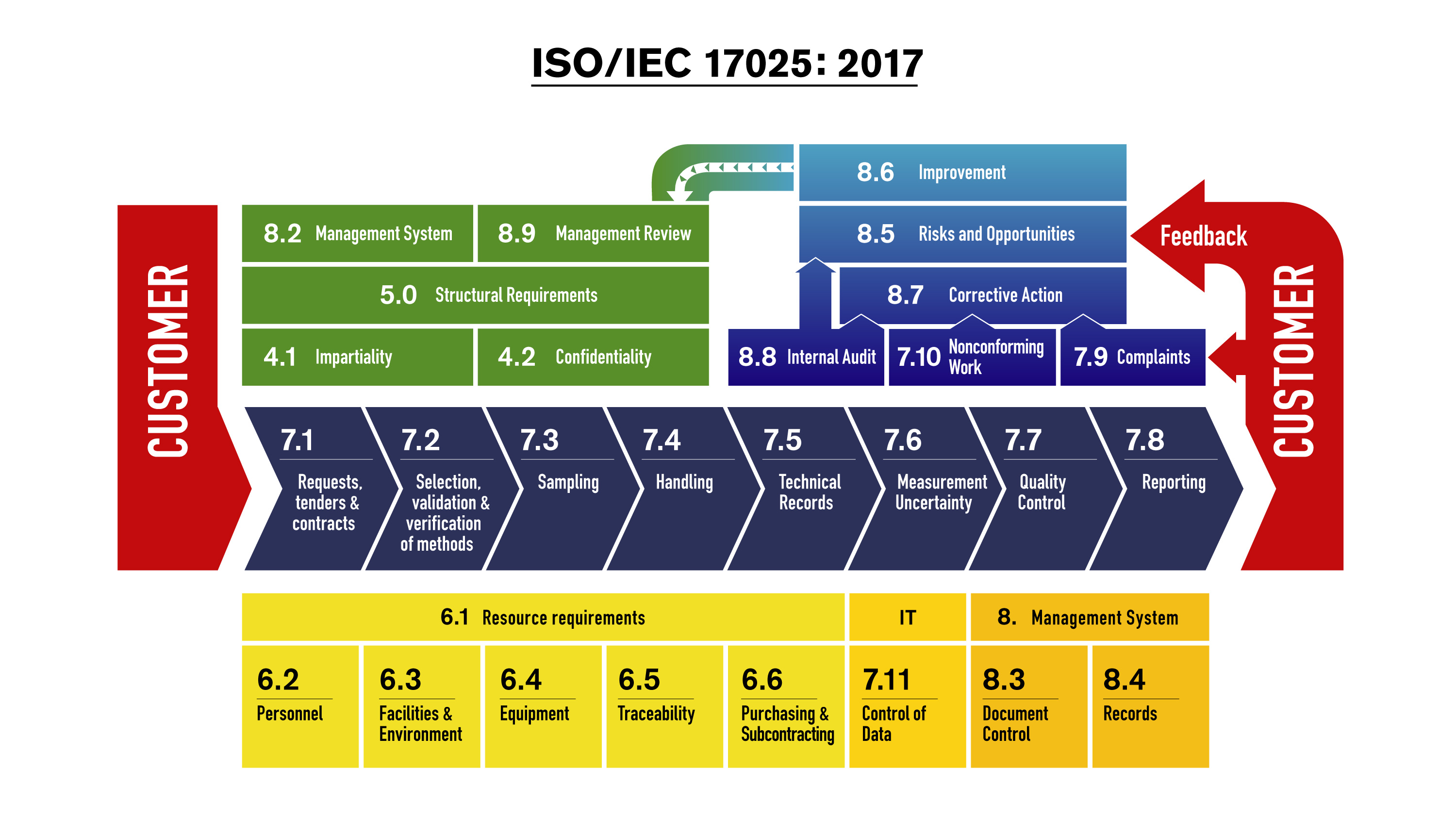 
Accreditation to this standard signifies a laboratory’s technical competency in producing accurate and reliable results. ISO 17025:2017 is an internationally recognized standard that outlines general requirements for testing and calibration laboratories’ competence, impartiality, and consistent operation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
